
For billions of years, microbes like bacteria have quietly transformed the Earth. They have re-routed the flow of nutrients around our planet, infused the atmosphere with oxygen, and built the biosphere from the bottom up. It is hard to overstate the palaeontological importance of “simple” single-celled micro-organisms. Unfortunately for palaeontologists, however, these small, soft, delicate forms of life are very difficult to study in the fossil record. Identifying and interpreting fossil bacteria is a formidable challenge, requiring close collaboration between geologists, microbiologists, and specialists in high-resolution chemical analysis. Nevertheless, it is a challenge we must accept if we want to understand where we came from, reconstruct Earth’s biosphere in the distant past, or seek evidence of life on Mars.
The most basic problem for microbial palaeontology is the difficulty of distinguishing between real microfossils and other geological phenomena that merely look like them. We know from painful experience that many non-biological processes in nature can produce life-like structures, especially at microscopic scales. Geologists have always struggled to distinguish these from real fossils, and the history of palaeontology is littered with such cases of mistaken identity, from the so-called “nanofossils” in a martian meteorite (amorphous mineral particles) to the world’s “oldest fossil cyanobacteria” (clay minerals coated in carbon).
In this context, I have been investigating strange, microscopic filaments commonly found in the hollows of ancient rocks — slender, curving threads that sprout grassily from the walls of pores and cracks. They look very much as though they were once alive but are composed of minerals, chiefly iron oxides (the same minerals that give Mars its rusty colour). These inorganic threads seem to be everywhere on Earth and to have been here a very long time. Geologists have found them on at least five continents and even beneath the Pacific Ocean. They are inside limestones, basaltic lavas, seafloor silica deposits, and deeper subseafloor rocks. Some filaments seem to have formed within a few metres of the Earth’s surface; others formed kilometres underground. The oldest examples are four billion years old; the youngest, less than a million. Palaeontologists have cautiously inferred that all of these are fossil bacteria or fungi. But are they?
Until recently, no non-biological processes seemed able to explain the iron-mineral filaments. They are not simply needle-like crystal fibres, as a team at Harvard proposed unconvincingly last year; indeed, they are composed of innumerable much smaller crystals. Neither are they “silica-carbonate biomorphs”, marvellous self-organised mineral growth structures well known to palaeontologists who work on microbes, thanks to the work of Juan Manuel García-Ruiz and his collaborators. These phenomena cannot explain the composition and structure of iron-mineral filaments. For example, the filaments have circular cross-sections (unlike crystals); some are even long hollow tubes, as if the iron minerals once precipitated upon living cells that simply died and rotted out. Some filaments branch off and reconnect with each other like fungi or actinobacteria weaving through soil (unlike the silica-carbonate biomorphs, which are also made of the wrong stuff). Some even contain complex carbon-based molecules (“organic matter”) that seem to be derived from living organisms; in at least one known case, these molecules include chitin, a signature of fungi.
If this evidence pointed to a biological origin for all the iron-mineral filaments in Earth’s rocks, then the implications for palaeontology would be enormous. The four-billion-year-old examples would be the oldest known fossils on Earth (as claimed by their discoverers), and some elaborately branched two-billion-year-old specimens would be our oldest plausible candidate for fossil fungi. The iron-mineral filaments would also provide a valuable record of the bacteria and fungi that live in the deep, dark recesses of Earth’s crust. This subsurface “deep biosphere” is a global subsurface microbial ecosystem that today stores huge amounts of carbon and may have hosted some of the first life on Earth. The notion that microbes living in deep, anaerobic, basaltic rocks have left behind a fossil record is especially exciting for astrobiologists looking for evidence of life in similar settings on Mars.
The palaeontological interpretation is not without difficulties, however. The vast majority of iron-mineral filaments contain no organic matter. Even if the few that do really are fossils, we can’t assume that all the other filaments have the same origin. And the morphological analogy to known forms of life is not always perfect, even accounting for the distorting effects of fossilization. Some filaments, for example, are suspiciously irregular or inconsistent in shape, or appear densely crowded around mineral particles from which they radiate outwards like crystal fans. These clues have led me to investigate a new hypothesis about their origin: that they are natural chemical gardens.
Chemical Gardens
Chemical gardens were discovered more than three hundred years ago, possibly by Johann Glauber, a Dutch alchemist who reported that metallic salt grains placed into waterglass (a silicate solution) seemed to germinate and send up shoots within hours, “in the form of plants and trees, each metal according to its inmost colour and property, which metalline vegetations are called philosophical trees, both pleasant to the eye and of good use.”1 On a modern understanding, these structures result from a chemical reaction between the dissolving “seed” salts and the surrounding solution. This reaction produces a gelatinous bag-like membrane that traps the dissolving metal solution inside. Osmotic inflow across the membrane forces it to rupture and extend, producing curving tubular structures that tend to grow upwards because of buoyancy (the exterior medium is denser than the interior). Ruptures in the tube walls generate branches, and an impressive host of lifelike forms results. Interestingly, metal oxide minerals then precipitate on the tubes, coating and partially filling them.
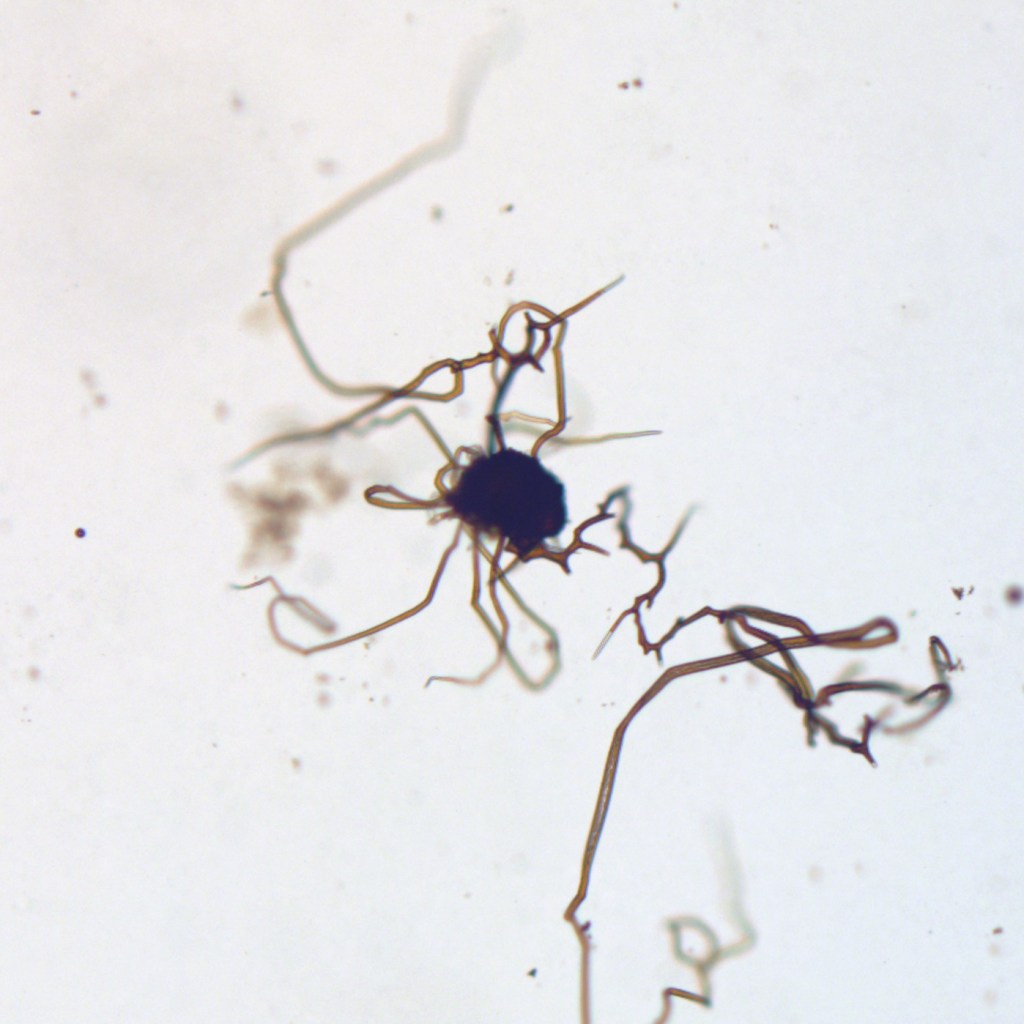
The idea that naturally occurring chemical gardens might look misleadingly like fossils is not new — it was suggested by J.E. Hawley2 in 1926, and it has resurfaced from time to time since then, including in a PhD thesis submitted to Trinity College Dublin in 2008, and in some papers by García-Ruiz. But the idea that the iron-mineral filaments I’ve described might be chemical gardens has never gathered much traction or been tested experimentally by palaeontologists. My contribution3 was to show that chemical gardens easily reproduce both the complex tubular, branching, re-connecting morphology and the iron-oxide composition of the naturally occurring iron-mineral filaments using ingredients and conditions that are probably not too far removed from some natural groundwaters. In fact, I was deeply surprised — disturbed, even — by how readily these non-biological reactions could mimic many features previously considered unique to life: so-called “criteria for biogenicity”.
My results don’t show that all iron-mineral filaments in rocks are non-biological in origin. It remains for future work to discover how true filamentous microfossils and chemical gardens can be distinguished from each other in nature. To achieve this, I suspect we must first explore what happens to bona fide microbial cells when they are encrusted by iron minerals in a naturalistic way — something that can and should be investigated in the lab. The fossil record is an extraordinary archive of life on Earth, but we will never be able to read it properly until we learn how it was written.
- Barge, L.M., Cardoso, S.S., Cartwright, J.H., Cooper, G.J., Cronin, L., De Wit, A., Doloboff, I.J., Escribano, B., Goldstein, R.E., Haudin, F. and Jones, D.E., 2015. From chemical gardens to chemobrionics. Chemical reviews, 115(16), pp.8652-8703.
- Hawley, J.E., 1926. An evaluation of the evidence of life in the Archean. The Journal of Geology, 34(5), pp.441-461.
- McMahon, S. 2019. Earth’s earliest and deepest purported fossils may be iron-mineralized chemical gardens. Proceedings of the Royal Society B, 286.

Leave a comment